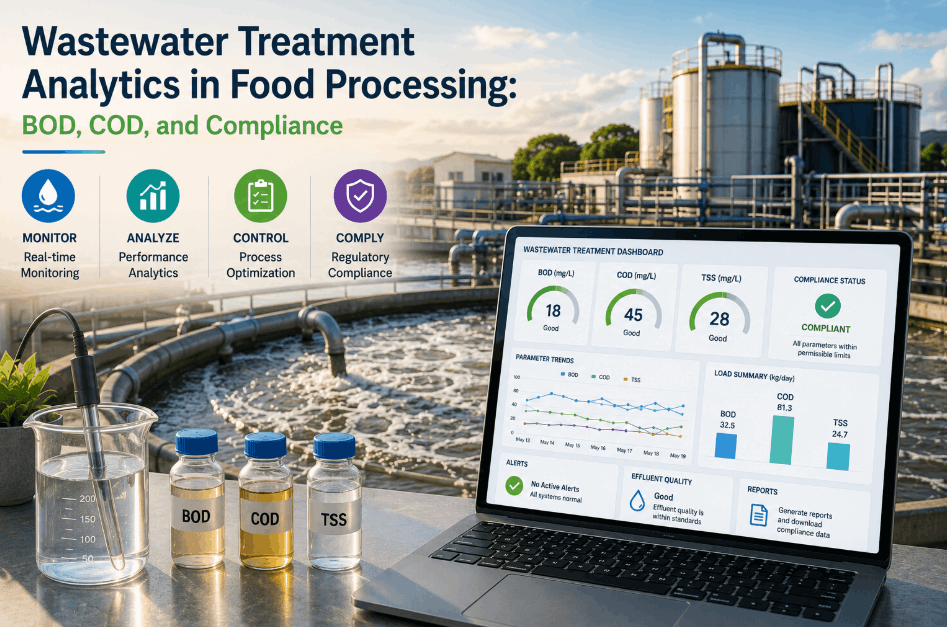
BRC Global Standard compliance has become one of the most demanding certification challenges for food manufacturers — not because the standard itself is unclear, but because the analytics documentation requirements it imposes across infrastructure programs, calibration systems, corrective action workflows, and audit trails are too granular and interconnected to manage manually at scale. Quality Assurance Directors leading multi-site food operations increasingly recognize that traditional documentation approaches create compliance gaps that only become visible during audits — and by then, the cost of remediation far exceeds the cost of prevention. AI-driven analytics platforms are now redefining how food manufacturers approach BRC audit preparation, transforming documentation from a reactive burden into a continuously maintained, audit-ready asset. If your facility is struggling to maintain consistent BRCGS compliance documentation across multiple production lines or sites, Book a Demo to see how AI-powered analytics closes the documentation gap before your next audit window.
What BRC Global Standard Compliance Actually Requires From Your Analytics Documentation
The BRCGS Food Safety Standard — currently in Issue 9 — requires food manufacturers to maintain documented evidence across every operational control point, not just at the product level. For Quality Assurance Directors, this means that BRC analytics documentation must span equipment calibration histories, environmental monitoring programs, supplier approval records, corrective and preventive action (CAPA) logs, internal audit trails, and performance trend data — all linked and traceable to specific production events. The challenge is not understanding what documentation is required; it is ensuring that documentation is generated consistently, stored accessibly, and presented coherently when an unannounced audit begins.
BRC compliance auditors evaluate documentation quality on two dimensions: completeness and traceability. Completeness means every required record exists for every required timeframe. Traceability means each record can be linked to the specific equipment, process, batch, or corrective action it governs. Facilities that rely on manual documentation systems — paper logs, disconnected spreadsheets, siloed ERP modules — consistently struggle on the traceability dimension, creating minor or major non-conformances that could be entirely eliminated through AI-driven BRC compliance documentation architecture.
Infrastructure Program Records
BRC requires documented evidence that infrastructure maintenance programs — pest control, cleaning schedules, facility inspection cycles — are executed consistently and that deviations are captured, investigated, and resolved with documented corrective actions. Manual tracking creates gaps that accumulate between audit cycles.
Calibration & Measurement Records
Every monitoring and measuring device used in a BRC-controlled process requires documented calibration records with traceability to national or international measurement standards. AI analytics platforms can automatically log calibration events, flag overdue instruments, and generate compliant calibration certificates without manual data entry.
Corrective Action Documentation
BRC Issue 9 places significant emphasis on the quality of corrective action documentation — requiring root cause analysis, immediate corrective actions, preventive actions, and effectiveness reviews to be formally documented for every non-conformance. Incomplete CAPA records are among the most common sources of audit findings across food manufacturing facilities.
Trend Analysis & KPI Reporting
BRCGS requires facilities to demonstrate ongoing performance monitoring through trend analysis of key quality and safety indicators. This demands structured analytics documentation that captures KPI trajectories over time — not just point-in-time snapshots — as evidence of continuous improvement program effectiveness.
How AI-Driven Analytics Generates BRC-Compliant Documentation Automatically
Modern BRC audit documentation requirements cannot be reliably met through human-managed systems alone — the volume, frequency, and interconnectedness of required records exceed what quality teams can maintain without systematic automation. AI-driven analytics platforms address this by structuring documentation generation directly into operational workflows, ensuring that every calibration event, every corrective action, every monitoring deviation automatically produces a BRC-compliant record without requiring a separate manual documentation step. Quality Assurance Directors who want to see this architecture in action can Book a Demo and review a live demonstration of automated BRCGS documentation generation mapped to their specific facility profile.
Automated Calibration Record Generation
AI platforms integrate with calibration management systems and sensor networks to automatically log every calibration event with timestamped evidence, equipment identifiers, reference standard traceability, and pass/fail outcomes. Overdue calibration alerts surface in real time, and calibration history reports are generated on demand in BRC-auditor-ready formats — eliminating the manual certificate compilation that typically consumes weeks of QA team time ahead of scheduled audits.
Inputs: Sensor Networks · CMMS · Calibration SchedulesCAPA Workflow Documentation Engine
Every non-conformance detected through automated monitoring — a temperature exceedance, a CCP deviation, a cleaning verification failure — immediately triggers a structured CAPA workflow that guides quality personnel through root cause analysis, corrective action documentation, preventive action planning, and effectiveness review. All workflow steps are time-stamped, user-attributed, and stored in a searchable repository that satisfies BRC's corrective action traceability requirements completely.
Inputs: Monitoring Alerts · Process Deviations · Quality EventsInfrastructure Program Compliance Tracking
Scheduled infrastructure programs — pest control visits, deep cleans, facility inspections, maintenance rounds — are tracked against planned frequencies with automated completion verification and deviation flagging. When a scheduled activity is overdue or completed outside specification, the system generates a documented deviation record automatically, ensuring BRC auditors see a complete and honest compliance history rather than a curated selection of successful completions.
Output: Completion Records · Deviation Logs · Program Trend ReportsAudit-Ready Report Generation
The platform synthesizes operational data, corrective action histories, calibration records, and KPI trend analyses into formatted audit report packages aligned to BRC clause structure — enabling QA Directors to provide auditors with pre-organized evidence packs rather than compiling documentation manually during the audit window. Report generation that previously required days of analyst effort is compressed to minutes through AI-driven document assembly.
Output: Clause-Mapped Reports · Evidence Packs · KPI Trend AnalysesBRC Analytics Documentation Requirements by Clause: What QA Directors Must Demonstrate
Understanding BRC analytics requirements at the clause level is essential for Quality Assurance Directors building compliant documentation systems. The table below maps the primary BRC Issue 9 clauses that carry analytics documentation obligations to the specific data types, record formats, and review frequencies they require — providing a structured framework for gap assessment against current documentation practices. Facilities ready to benchmark their current documentation coverage against full BRCGS compliance requirements can Book a Demo for a structured gap assessment with our food safety analytics team.
| BRC Issue 9 Clause | Documentation Requirement | Analytics Data Type | Review Frequency | AI Automation Opportunity |
|---|---|---|---|---|
| Clause 4.3 — HACCP | CCP monitoring records, deviation logs, corrective action evidence | Real-time sensor data, process parameter logs | Continuous / Per Batch | Automated CCP monitoring with instant deviation capture |
| Clause 6.1 — Equipment Maintenance | Planned maintenance schedules, completion records, backlog reports | CMMS work order data, OEE history | Weekly / Monthly | Predictive maintenance scheduling with auto-generated work orders |
| Clause 6.2 — Calibration | Calibration certificates, instrument registers, traceability records | Calibration event logs, measurement uncertainty data | Per Calibration Event | Automated calibration record generation and overdue alerts |
| Clause 3.7 — Corrective Action | Non-conformance records, root cause analyses, CAPA effectiveness reviews | Quality event data, process deviation logs | Per Non-Conformance Event | Structured CAPA workflow with automated documentation capture |
| Clause 3.11 — Internal Audits | Internal audit schedules, completed audit reports, finding closure evidence | Audit finding data, corrective action completion rates | Minimum Annual (risk-based) | AI-generated audit reports with clause-mapped finding summaries |
| Clause 4.15 — Traceability | Batch traceability records, mass balance documentation, recall test results | Production batch data, ingredient lot records | Per Batch / Annual Test | Automated traceability chain mapping with instant recall simulation |
| Clause 6.4 — Pest Control | Pest control visit records, bait station maps, trend analysis reports | Pest activity data, contractor visit logs | Per Visit / Quarterly Trend | Automated activity trend analysis with escalation alerting |
Key Analytics Capabilities That Drive BRC Food Safety Standard Compliance
Achieving and maintaining BRC food safety standard certification requires more than point-in-time documentation — it demands a continuous analytics infrastructure that monitors compliance status across all clauses simultaneously, surfaces emerging gaps before they become audit findings, and generates the structured evidence records that auditors require to confirm compliance with confidence. The following capabilities represent the analytics foundation that enables QA Directors to lead a genuinely audit-ready operation rather than an audit-preparation exercise. Organizations evaluating their current analytics infrastructure against BRCGS requirements can Book a Demo for a capability demonstration mapped to specific BRC clause obligations.
Real-Time CCP & OPRP Monitoring Analytics
Continuous sensor-driven monitoring of all critical control points and operational prerequisite programs with automated limit breach detection, instant alert routing, and timestamped deviation records that satisfy BRC's CCP documentation requirements without manual data entry at the production line level.
Automated Instrument Calibration Tracking
Complete instrument register management with calibration due date tracking, overdue escalation alerts, automated certificate generation, and traceability chain documentation — ensuring calibration compliance is maintained continuously and presented in BRC-compliant format during unannounced audits.
Structured Corrective Action & Root Cause Analytics
AI-guided CAPA workflows that enforce BRC-compliant root cause analysis methodologies, document immediate and preventive actions with user attribution, schedule effectiveness reviews, and generate clause-referenced CAPA summary reports that demonstrate systematic non-conformance management to auditors.
Quality KPI Trend & Continuous Improvement Analytics
Statistical trend analysis of defect rates, non-conformance frequencies, audit finding categories, and CAPA closure times — generating the continuous improvement evidence that BRC Issue 9 requires facilities to demonstrate as proof that their food safety management system is driving genuine operational improvement, not just generating records.
Supplier Approval & Performance Analytics
Centralized supplier approval status tracking, certificate of conformance monitoring, supplier audit scheduling, and performance trend analysis — ensuring BRC's supplier management clause requirements are met with a documented, risk-scored supplier portfolio that auditors can review comprehensively in minutes.
AI-Generated Internal Audit Report Documentation
AI-driven internal audit support tools that structure findings against BRC clause references, generate formatted audit reports with finding severity classifications, track corrective action commitment dates, and produce audit program effectiveness analyses that satisfy BRC's internal audit documentation requirements completely.
AI-Driven BRC Audit Preparation: From Documentation Gap to Audit-Ready Evidence in Hours
The traditional BRC audit preparation cycle — weeks of document retrieval, spreadsheet compilation, record verification, and evidence packaging — represents one of the most resource-intensive recurring burdens on food manufacturing quality teams. AI-driven analytics platforms fundamentally change this workflow by maintaining audit-ready documentation continuously rather than assembling it episodically. The following workflow illustrates how QA Directors using AI analytics move from documentation baseline to fully prepared audit evidence package without the manual scramble that characterizes conventional BRC audit preparation.
Continuous Compliance Status Monitoring
The AI platform continuously scores compliance status across all BRC clauses with analytics documentation requirements — calibration coverage, CAPA closure rates, infrastructure program completion percentages, and monitoring record completeness. QA Directors see a live compliance dashboard rather than discovering gaps during pre-audit document reviews, enabling proactive remediation on a rolling basis throughout the certification cycle.
Automated Documentation Gap Detection
When the platform identifies documentation gaps — missing calibration records for instruments approaching their due date, CAPA records without completed effectiveness reviews, or infrastructure program completions not logged within the required window — it generates automated alerts with clause references and remediation timelines, ensuring QA teams address gaps on their own schedule rather than an auditor's timeline.
Clause-Mapped Evidence Package Assembly
When an audit is scheduled — or announced without warning — the platform generates a structured evidence package that maps all relevant documentation to the specific BRC clause it satisfies. Calibration certificates, CAPA records, monitoring logs, trend analyses, and internal audit reports are compiled into an auditor-navigable format that demonstrates comprehensive compliance coverage without requiring manual document retrieval from multiple disconnected systems.
Post-Audit Finding Integration & Closure Tracking
Non-conformances and observations raised during BRC audits are entered directly into the platform's CAPA management system, where corrective action commitments, target closure dates, and responsible owners are tracked automatically. The platform monitors closure progress and generates escalation alerts for overdue actions — ensuring the corrective action response to audit findings is as well-documented as the original compliance evidence that preceded them.
Measurable Outcomes: What AI-Driven BRC Compliance Analytics Delivers for QA Operations
For QA Directors building the business case for AI-driven compliance analytics investment, the value proposition extends well beyond audit outcome improvement. The operational efficiency gains from eliminating manual documentation workflows, reducing pre-audit preparation cycles, and achieving continuous compliance monitoring create measurable returns across the entire quality management function — not just at certification renewal time.
Reduction in minor and major non-conformances at BRC audits, driven by continuous documentation gap detection and proactive remediation before audit windows open.
Reduction in QA team time spent on pre-audit documentation preparation, through automated evidence package assembly replacing manual document compilation processes.
Improvement in on-time CAPA closure rates, enabled by automated deadline tracking and escalation alerting that prevents corrective actions from stalling without visibility.
Calibration compliance coverage achieved across all monitored instruments, through automated scheduling and overdue alerting that eliminates the missed calibrations that create BRC findings.
Why QA Directors Choose AI Analytics for BRCGS Compliance Documentation
The cost of BRC non-conformances extends far beyond the audit remediation fees and certification delays they create — a major finding in a high-volume food category can trigger retailer delisting, production suspension, and brand reputation damage that dwarfs any technology investment in compliance infrastructure. AI-driven analytics platforms that eliminate documentation gaps, automate calibration tracking, and generate structured CAPA evidence represent a risk management investment that pays for itself many times over in the first certification cycle alone. QA Directors ready to quantify the compliance risk exposure in their current documentation system can Book a Demo for a live demonstration of automated BRC compliance documentation generation tailored to their specific facility's clause requirements and risk profile.
BRC Global Standard Compliance Analytics — Frequently Asked Questions
How does AI analytics documentation differ from manual BRC record-keeping systems?
Manual systems capture documentation after operational events occur, often with delays and omissions that create traceability gaps. AI analytics platforms generate documentation at the moment of the operational event — a calibration, a CCP deviation, a maintenance completion — ensuring records are complete, timestamped, and immediately searchable without requiring separate documentation actions from quality or operations personnel.
Can AI-generated documentation satisfy BRC Issue 9 auditor requirements for record authenticity?
Yes. AI-generated documentation platforms produce records with user attribution, system timestamps, and audit trail logs that demonstrate record authenticity and integrity — requirements that BRC Issue 9 applies equally to electronic documentation systems. Platforms operating under 21 CFR Part 11 or equivalent electronic record principles are specifically designed to meet these authenticity requirements.
Which BRC clauses create the highest documentation burden for multi-site food manufacturers?
Clauses governing HACCP monitoring (4.3), equipment calibration (6.2), corrective actions (3.7), and internal audit programs (3.11) consistently generate the highest documentation volume and complexity — particularly for multi-site operations where documentation must be maintained consistently across facilities with different equipment profiles, production processes, and local regulatory overlays.
How quickly can an AI analytics platform be deployed to support an upcoming BRC audit?
Cloud-native analytics platforms with pre-built ERP and CMMS connectors can be operational at a single facility within two to four weeks of deployment initiation, with initial compliance dashboards and CAPA workflows available within the first week of data integration. Historical data backfill from existing systems ensures the platform reflects the full documentation history required for certification review, not just activity from the deployment date forward.
Does AI analytics software support BRC unannounced audit requirements under Issue 9?
Directly. BRC Issue 9 introduced unannounced audit options that remove the preparation window that facilities previously relied on to organize documentation. AI analytics platforms that maintain continuous, audit-ready documentation eliminate the dependency on preparation time entirely — the evidence package that would take a quality team days to assemble manually is generated in minutes from the live compliance database when an unannounced auditor arrives on site.
Can AI-driven compliance analytics integrate with existing BRC-related quality management systems?
Yes. Enterprise analytics platforms use pre-built API connectors to integrate with major QMS, CMMS, ERP, and LIMS systems already deployed in food manufacturing environments — consolidating documentation from existing systems into a unified compliance intelligence layer rather than replacing infrastructure that quality teams already depend on for operational management.