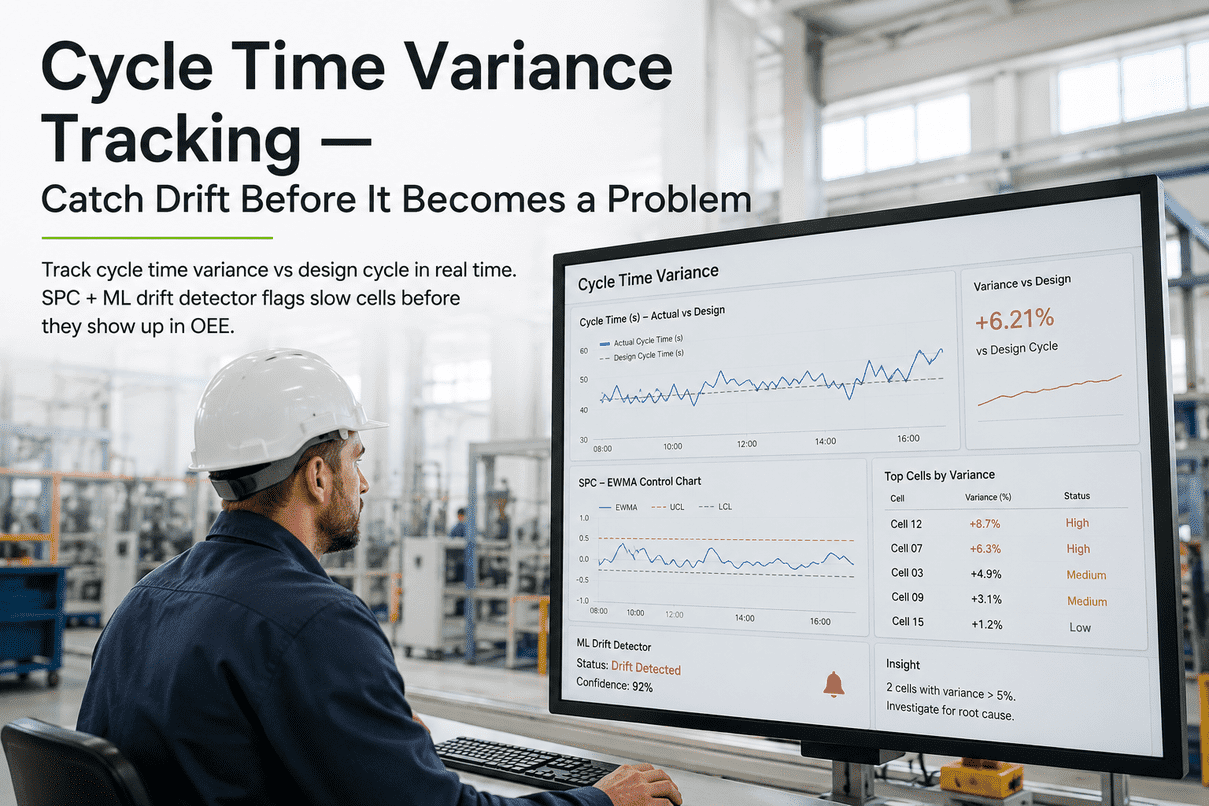
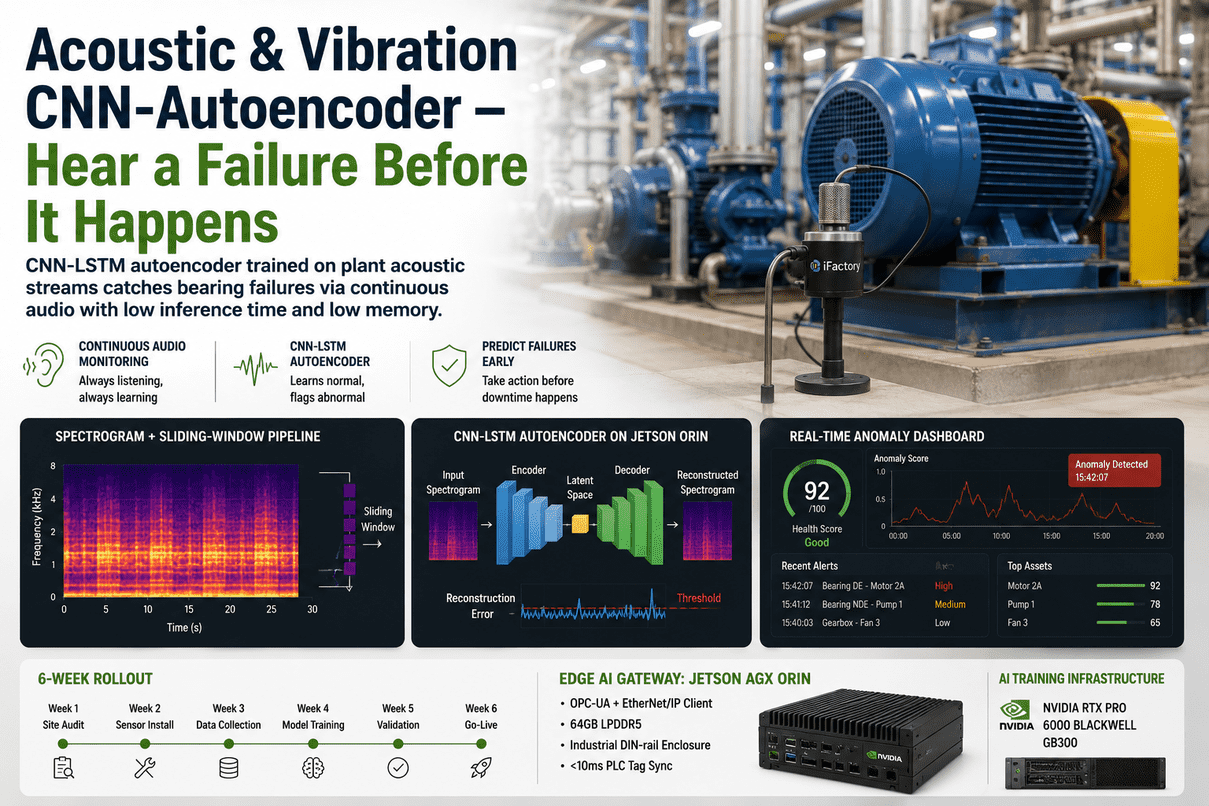
An API drug substance plant is a chemistry factory operating under a microscope. Every reactor charge, every crystallization endpoint, every impurity peak in your IPC HPLC carries downstream consequences — yield, polymorph stability, nitrosamine risk, ICH Q11 compliance. iFactory's API & Drug Substance AI Platform sits across this entire chemistry workflow — from raw material qualification, through reaction monitoring and crystallization endpoint detection, all the way to milling and packaging — using PAT-grounded chemometrics, reaction kinetics ML, and a plant LLM for batch genealogy and deviation drafting. Deployed on-prem on NVIDIA GB300 + H200 + Jetson, validated under GAMP 5, and aligned to ICH Q11 development principles.
Upcoming iFactory AI Live Webinar:
API & Drug Substance AI — Synthesis to DS Plant
Join our pharma chemistry team for a live walk-through of an AI platform deployed across small-molecule API and DS plants. Reaction monitoring, crystallization endpoint, impurity prediction, flow chemistry control, and electronic batch records aligned to ICH Q11 — built on 1,000+ enterprise implementations.
Why API Plants Are the Hardest Place to Deploy AI
Drug substance manufacturing is high-temperature, high-pressure, multi-step organic chemistry running on regulated infrastructure. The data is messy — IPC HPLCs come back hours late, reactor logs are in batch sheets, and crystallizer scale-up is half art. Most AI platforms can't even ingest this data, let alone reason about it. Schedule a chemistry-readiness review and we'll model your synthetic route into the AI pipeline.
Reaction completion checked by IPC HPLC every 2 hours. Operators sit on completed reactions burning energy and risking impurity formation while waiting for QC results.
Polymorph control, particle size distribution, and yield drift between campaigns. Lab-to-plant scale-up loses 5–15% yield to seed-handling and cooling-rate variability.
Risk emerges 6–12 months after market launch, after recalls and warning letters. Pre-emptive impurity prediction across the synthetic route is still rare in industry.
Trace one starting material lot across 14 intermediates and 3 dosage forms? That's a 6-week investigation. Regulators expect it in 48 hours.
AI Across Every Step of Your Synthetic Route
Drug substance is a sequence — raw materials become intermediates become API. Every step generates spectra, kinetics, and IPC data. Our platform deploys a model layer that follows the molecule.
NIR/Raman incoming inspection, supplier-lot variability scoring, CoA digitization via OCR + LLM.
In-situ Raman + ReactIR feeds a kinetics ML model. Endpoint called automatically — reaction stops at peak conversion, before degradation begins.
Phase-cut detection from inline conductivity, layer interface vision AI, solvent recovery efficiency tracked across campaigns.
FBRM + PVM data into a Gaussian Process model predicts PSD and polymorph form. Cooling profile auto-adjusted to land target d50.
Cake resistance, residual solvent prediction, drying endpoint via NIR + LSTM. Eliminates over-drying and energy waste.
PSD prediction post-mill, electrostatic risk modeling, batch release prediction tied to formulation downstream needs.
Inside the Reactor — From Spectra to Endpoint in Real Time
A typical pilot or commercial reactor runs blind for 80% of its cycle. Operators rely on temperature, jacket Δ, and scheduled IPC pulls. We replace that with continuous spectroscopic intelligence.
Raman, ReactIR, FTIR, FBRM probes stream spectra every 30 seconds. Kafka pipeline lands data in the on-prem AI engine within 200 ms.
Spectra → starting material concentration → conversion %. Model validated against historical batch HPLC data using tagged event windows.
When conversion plateau detected and side-reaction signal stays below threshold, AI calls endpoint. DCS gets the signal — operator confirms, batch advances.
Same spectra feed an impurity classifier — flags nitrosamine precursors, genotox, dimer formation before workup. QbD design-space stays intact.
Crystallization — The Make-or-Break Step, Now Predictable
Crystallization controls polymorph, PSD, residual solvent, and downstream filtration. It also reliably blows up at scale-up. Our Gaussian Process model fuses FBRM, PVM, temperature, and supersaturation data into a real-time controller. Talk to our crystallization team for a model-fit assessment on your API.
Continuous Flow & FDA AMT — AI That Comes Standard
FDA's Advanced Manufacturing Technology (AMT) program has accelerated continuous flow API adoption — but flow plants generate 50× the data of batch and demand sub-second control. Our platform was built for it.
Edge inference on Jetson Orin keeps mass flow controllers, temperature, and pressure within design space at every reactor stage of the flow train.
Raman, NMR, IR, GC sensors at every reactor stage feed a unified model. Out-of-spec material auto-diverted to waste — never reaches the next unit.
Bayesian optimization explores temperature, residence time, and stoichiometry within the design space — converges on optimum in 20–30 batches.
Multi-step flow trains share state. The platform predicts how perturbations in step 1 propagate to step 4 — closing the loop end to end.
ICH Q11, Q14, GAMP 5 & 21 CFR Part 11 — All Wired In
DS plants live under ICH Q11 (development & manufacture of drug substances) and Q14 (analytical procedure development). Add 21 CFR Part 11 audit trails and GAMP 5 categorization, and most AI tools fall apart. Ours start there.
The Compute Stack for a DS Plant
A typical multi-stage API plant lands on a tiered NVIDIA stack — Jetson at every reactor for sub-second control, H200 for chemometric model training, and a GB300 NVL72 for the plant LLM and multi-batch digital twin.
| Tier | Hardware | Workload | Latency | Deployment |
|---|---|---|---|---|
| Edge | NVIDIA Jetson Orin | Endpoint detection, vision QC, flow control | <30 ms | One per reactor / line |
| Plant | NVIDIA H200 (8-GPU) | PLS, GP, CNN training; multi-batch analytics | seconds | 2–4 nodes per facility |
| Core | NVIDIA GB300 NVL72 | Plant LLM, batch genealogy, deviation drafting | real-time inference | One rack per enterprise |
| Storage | Validated data spine | PAT raw spectra, batch history, audit trail | n/a | On-prem, GxP-validated |
Where the LLM Helps — and Where It Stays Out of the Loop
In a DS plant, an LLM is a powerful assistant — but never the process controller. Reaction kinetics, crystallization, and endpoint calling are governed by deterministic ML (PLS, PCA, GP, kinetics models). The LLM handles language-heavy tasks where it actually excels.
- Batch genealogy queries — "Trace SM lot SM-2247 across all derivatives"
- Deviation report drafting from raw event logs
- OOS investigation — surfacing similar past cases
- SOP retrieval, change control summaries
- Regulator question pre-drafting (FDA 483 response prep)
- Reaction endpoint calling (PLS + kinetics ML)
- Crystallization PSD & polymorph (GP + CNN)
- Impurity prediction (PLS classifier)
- Drying endpoint (NIR PLS + LSTM)
- All process control loops touching CQAs
From PFD to Production AI in 14–18 Weeks
A typical small-molecule DS deployment runs 14–18 weeks. The schedule below is a single-product, single-route example; multi-product sites stack additional cycles. Schedule a deployment-readiness review to get a timeline mapped to your specific synthetic route.
What Process Chemistry & QA Leaders Ask First
Both. The platform was built batch-first. Most of our deployed reaction endpoint and crystallization models are on stirred-tank batch reactors. Flow chemistry just gets a denser PAT footprint and tighter control loop.
No. The AI runs in advisory mode first — predictions logged, not enforced. After PQ confirms accuracy against your IPCs, the closed-loop modules get individually validated. Existing ICH Q11 control strategy is preserved, then extended.
The classifier is trained on your historical HPLC traces tagged with reaction conditions and SM lot data. Once a nitrosamine precursor pattern is detected, the model flags it before workup — when remediation is still possible. ICH M7 aligned.
EBRs stay where they are — Werum, Tulip, or in-house systems. The platform reads through a validated bridge and writes back AI predictions as new datapoints. No EBR re-implementation, no re-validation of your MES.
Get an AI Plan for Your Drug Substance Plant
Thirty minutes with our pharma chemistry engineers. Bring your synthetic route, current PAT footprint, and ICH Q11 control strategy. We'll show you exactly which AI modules apply, what compliance evidence we generate, and how the platform lands inside your validated state without disrupting production.