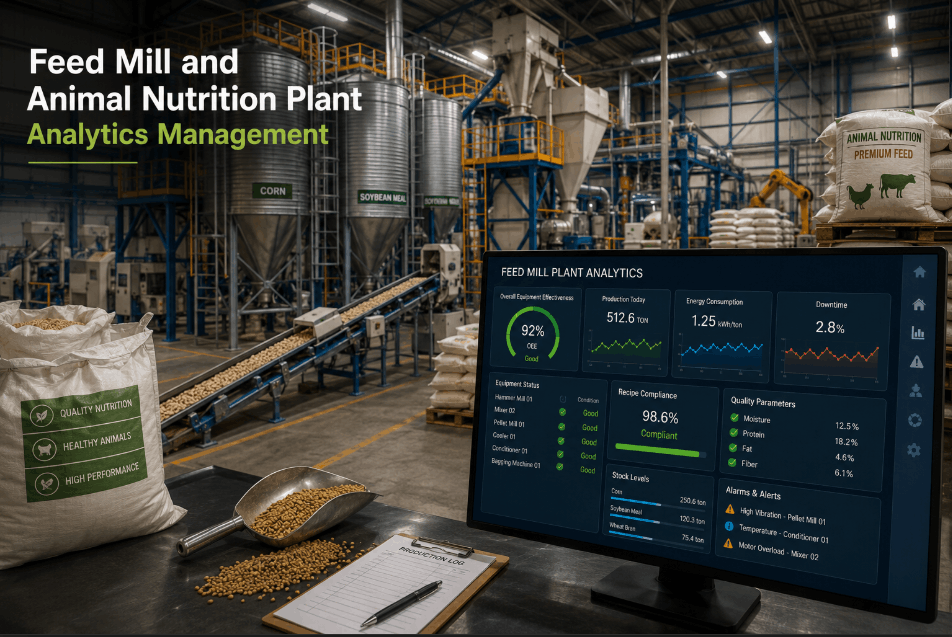
Feed mills and animal nutrition plants operate in one of the most compliance-intensive environments in food manufacturing — where equipment reliability, ingredient traceability, and FDA medicated feed compliance must run in parallel, across every shift, every line, every batch. Yet most feed production facilities still depend on manual inspection cycles, paper-based maintenance logs, and reactive repair workflows that leave pellet mills, extruders, mixers, and cooling systems vulnerable to unplanned downtime, regulatory exposure, and yield loss. Book a demo to see how ifactory's AI-driven analytics platform transforms feed mill compliance and equipment uptime across every critical asset in your facility.
The Hidden Cost of Reactive Maintenance in Animal Feed Manufacturing
Animal feed manufacturing operates on tight production windows, ingredient-sensitive processes, and strict regulatory timelines. A single unplanned pellet mill shutdown can cascade into missed batching schedules, ingredient segregation failures, and — in medicated feed environments — compliance documentation gaps that trigger FDA corrective action. Yet the majority of feed production facilities manage equipment health reactively: waiting for a die to seize, a mixer gearbox to fail, or a cooler to underperform before scheduling intervention.
Feed mill analytics software changes the operational model from reactive to predictive. By continuously monitoring vibration signatures, temperature profiles, motor load trends, and throughput deviations across pellet mills, extruders, mixers, and cooling systems, AI-driven platforms like ifactory surface failure precursors days or weeks before they become production stoppages. The result is a feed plant where maintenance is planned, not forced — and where compliance documentation is generated continuously, not reconstructed under audit pressure.
Feed Mill Equipment Analytics: From Pellet Mills to Cooling Systems
Effective feed plant analytics management requires monitoring coverage across every asset class in the production sequence — not just primary processing equipment. ifactory's preventive analytics platform maps to the complete feed manufacturing equipment portfolio, delivering asset-specific health monitoring, PM scheduling, and compliance documentation across each critical system. Facilities looking to reduce pellet mill downtime and extruder maintenance costs can book a demo to see equipment-level coverage in action.
Pellet mills are the highest-value and highest-risk assets in most feed production facilities. Die wear, roll adjustment drift, main shaft bearing degradation, and conditioner steam pressure deviations are leading causes of unplanned shutdowns — all of which generate measurable performance signals before failure. ifactory monitors pellet mill motor amperage trends, roll-to-die pressure differentials, throughput rate deviations, and conditioner temperature profiles in real time. AI-driven anomaly detection alerts maintenance teams to developing wear patterns and out-of-spec operating conditions before they escalate to production stoppages, extending die and roll life while protecting batch quality.
Feed extruders — particularly in aquafeed and pet food applications — operate under high mechanical stress and thermal load conditions that accelerate screw, barrel, and die insert wear. Maintaining consistent expansion ratios, bulk density targets, and moisture content at the extruder exit requires precise control over screw speed, barrel temperature zones, and moisture injection rates. ifactory tracks all critical extruder process parameters against specification ranges, triggering alerts when any variable drifts toward non-conformance. Screw wear progression is trended over time, enabling planned replacement scheduling aligned to production windows rather than emergency shutdowns.
Feed mixer performance directly determines the nutritional uniformity and medicated feed compliance of every batch produced. A mixer operating with worn ribbon flights, out-of-balance paddle arrangements, or incorrect mixing cycles produces batches with coefficient of variation (CV) values outside acceptable limits — which in medicated feed environments constitutes a direct regulatory non-compliance event. ifactory monitors mixer motor load signatures, mixing cycle duration adherence, and door seal integrity indicators in real time. Integration with batch management systems enables automatic CV trend reporting and flags any production run where mixing parameters deviate from the approved formula specification. Animal nutrition facilities can book a demo to see how mixer analytics integrates with medicated feed batch controls.
Feed cooling systems — counter-flow coolers, cross-flow coolers, and rotary coolers — are critical quality control points where finished pellet moisture content, temperature, and hardness are finalized before packaging. Inadequate cooling produces warm, high-moisture product vulnerable to mold growth and storage degradation. Overcooling creates excessive fines and increases packaging weight variability. ifactory tracks cooler inlet and outlet temperature differentials, airflow volume and static pressure, and residence time against production rate targets — alerting when cooling performance deviates from specification before product quality is compromised.
FDA Medicated Feed Compliance Tracking: From Manual Logs to Real-Time Documentation
FDA medicated feed manufacturing regulations — governed by the Current Good Manufacturing Practice (CGMP) requirements under 21 CFR Part 225 — impose documentation standards that manual systems structurally cannot meet at the speed and scale of modern feed production. Medicated premix lot traceability, mixer cleaning and flush verification between medication changes, label reconciliation, and batch record completeness are all subject to FDA inspection, and deficiencies in any of these areas can result in warning letters, facility consent decrees, or market withdrawals.
Key Medicated Feed Compliance Capabilities in ifactory
Feed Plant AI-Driven Analytics: What Your Maintenance Dashboard Reveals
The operational value of feed mill analytics software is only realized when the data it collects is surfaced to the right people, in the right format, at the right time. ifactory's compliance and analytics dashboards are designed around the actual decision workflows of feed plant quality managers, maintenance supervisors, and operations directors — delivering facility-level and asset-level visibility without requiring data science expertise to interpret.
| Dashboard View | Primary Users | Key Metrics Surfaced | Decision Enabled |
|---|---|---|---|
| Equipment Health Overview | Maintenance Supervisor | Asset health scores, open alerts, PM compliance rate | Daily maintenance prioritization |
| Pellet Mill Performance | Production Manager | Throughput trends, die wear index, motor load history | Die replacement scheduling, throughput optimization |
| Medicated Feed Compliance | Quality Director | Batch record completeness, open flush verifications, traceability chain status | FDA audit readiness, corrective action prioritization |
| Facility Compliance Risk Score | Plant Manager | Aggregate compliance risk by line, shift, and asset class | Resource allocation, audit preparation timing |
| PM Completion Tracking | Maintenance Planner | On-time PM rate, overdue tasks, upcoming scheduled events | Work order planning, parts procurement |
| Ingredient Segregation Status | Quality Assurance | Active segregation deviations, routing alerts, hold status | Cross-contamination prevention, lot disposition |
Deploying Feed Mill Analytics Software: What the Implementation Process Looks Like
One of the most common barriers to adopting feed plant AI-driven analytics is uncertainty about deployment complexity — particularly for facilities with legacy equipment, multiple production lines, and existing ERP or batch management systems. ifactory's implementation methodology is designed specifically for the operational realities of animal feed manufacturing environments, with phased deployment, risk-tiered rollout sequencing, and integration capability with existing production infrastructure.
All production assets — pellet mills, extruders, mixers, coolers, hammer mills, conveyors, and ancillary systems — mapped into ifactory's asset management framework. Medicated feed compliance control points configured against 21 CFR Part 225 requirements. Existing PM schedules and OEM maintenance intervals migrated from paper or spreadsheet systems into the digital PM module.
Deployment sequenced by production risk tier: primary pellet mill lines and medicated feed production equipment activated first. Real-time monitoring and compliance documentation begin accruing immediately on priority assets. Quality team training integrated into each activation window to ensure operational adoption without production disruption.
ifactory's data layer integrates with existing ERP, batch management, and receiving systems — creating unified ingredient traceability and batch record data chains. AI anomaly detection engine trained on facility-specific equipment performance baselines, establishing deviation thresholds calibrated to actual operating conditions rather than generic industry benchmarks.
All production lines and equipment assets active under unified compliance monitoring. First complete FDA-format medicated feed audit documentation package generated and reviewed by quality director. Audit export templates validated against regulatory requirements before the first inspection cycle. No operational interruptions during installation.
Why AI-Driven Feed Mill Analytics Delivers Compounding Operational Returns
Predictive failure detection on pellet mill assets reduces unplanned downtime by identifying wear signatures 24–72 hours before shutdown. The primary drivers of pellet mill failure — die wear progression, main bearing degradation, and conditioner performance decline — all generate detectable signal patterns well before catastrophic failure. AI-driven monitoring converts these signals into advance alerts that enable planned maintenance scheduling, eliminating the emergency repair cycles that dominate reactive maintenance cost structures in animal feed manufacturing.
Digital medicated feed compliance documentation eliminates the structural vulnerability of paper-based FDA audit preparation. FDA inspectors reviewing medicated feed manufacturing records under 21 CFR Part 225 are specifically trained to identify retroactively completed documentation — and manual systems structurally produce retroactive records. ifactory's real-time batch record generation and mixer verification logging eliminates retroactive documentation by design, producing audit-ready records at the point of production rather than weeks later under inspection pressure.
Unified equipment analytics and compliance tracking eliminates the data silos that create audit exposure in multi-line feed facilities. In facilities where maintenance records, batch documentation, and compliance logs are maintained in separate systems — or on paper — connecting equipment performance data to compliance outcomes requires manual correlation that is time-consuming and error-prone. ifactory's unified platform links equipment health data to production events and compliance records automatically, enabling quality teams to trace any compliance deviation back to its equipment-level root cause.
Animal feed manufacturing plants that deploy AI-driven analytics consistently recover platform investment within the first full certification cycle through documentation labor savings and downtime reduction. The financial return on feed mill analytics software is driven by three compounding savings streams: elimination of emergency maintenance labor and parts costs, reduction of compliance documentation labor hours, and prevention of production losses from unplanned equipment shutdowns. Facilities with active medicated feed compliance obligations add a fourth stream: avoidance of FDA corrective action and recall costs. Feed plant managers can book a demo to model ROI against their facility's current maintenance and compliance cost baseline.
Feed Mill Analytics Management: Building a Compliance-Ready, Uptime-Optimized Animal Nutrition Plant
The operational challenges facing feed mills and animal nutrition plants — unplanned equipment downtime, medicated feed compliance exposure, documentation gaps, and the compounding labor burden of manual maintenance tracking — are not inevitable features of feed manufacturing. They are the predictable outcomes of a compliance infrastructure that has not kept pace with the complexity of modern feed production. AI-driven analytics platforms like ifactory close that gap by delivering continuous equipment health monitoring, real-time compliance documentation, and audit-ready record generation across every critical asset in the feed production sequence.
Feed facilities that deploy preventive analytics across their pellet mills, extruders, mixers, and cooling systems are not simply reducing maintenance costs — they are building the operational foundation for sustainable certification compliance, retailer and customer confidence, and the kind of production predictability that enables growth. The technology exists, the deployment timeline is proven, and the ROI case is clear. The question is not whether AI-driven feed mill analytics delivers value — it is how much longer your facility can afford to operate without it.